Lamina®



Case Reports
Publications
Videos
- Tissue origin :
Cortical bone - Tissue collagen :
Preserved - Physical form :
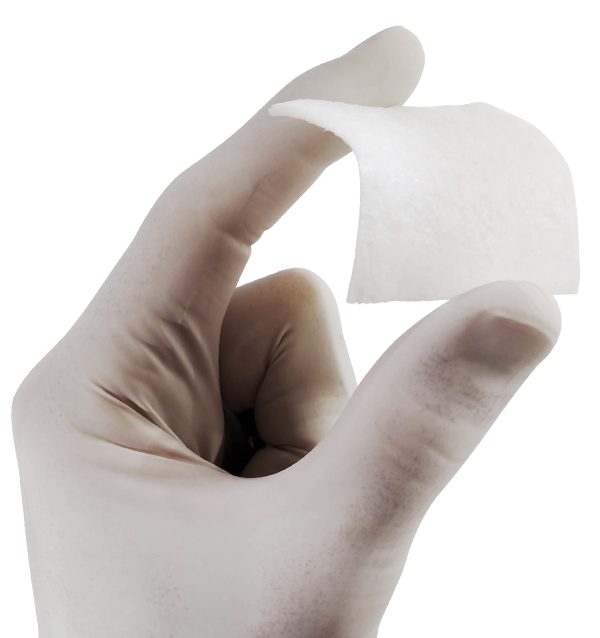
Lamina soft: semi-rigid flexible dried lamina
Lamina: rigid dried lamina, flexible after re-hydration - Composition :
100% cortical bone - Thickness :
0.5 mm (±0.1 mm)
0.7 mm (±0.1 mm)
1.0 mm (±0.1 mm)
3 mm (±1 mm) - Packaging :
0.5 mm: 25x25 mm, 25x35 mm (oval)
0.7 mm: 35x15 mm
1.0 mm: 35x35 mm (Curved), 20x40 mm
3.0 mm: 30x30 mm - Product codes :
LS25FS | 25x25 mm | 0.5 mm | soft | Porcine
LS25FE | 25x25 mm | 0.5 mm | soft | Equine
LS23FS | 25x35 mm (Oval) | 0.5 mm | soft | Porcine
LS23FE | 25x35 mm (Oval) | 0.5 mm | soft | Equine
LS24LS | 20x40 mm | 1.0 mm | soft | Porcine
LS10HS | 35x35 mm (Curved) | 1.0 mm | soft | Porcine
LS10HE | 35x35 mm (Curved) | 1.0 mm | soft | Equine
LS03SS | 30x30 mm | 3.0 mm | soft | Porcine
LS03SE | 30x30 mm | 3.0 mm | soft | Equine
LS15LS | 35x15 mm | 0.7 mm | rigid | Porcine
LS35LS | 35x35 mm | 1.0 mm | rigid | Porcine - GMDN code : 46425
- Characteristics
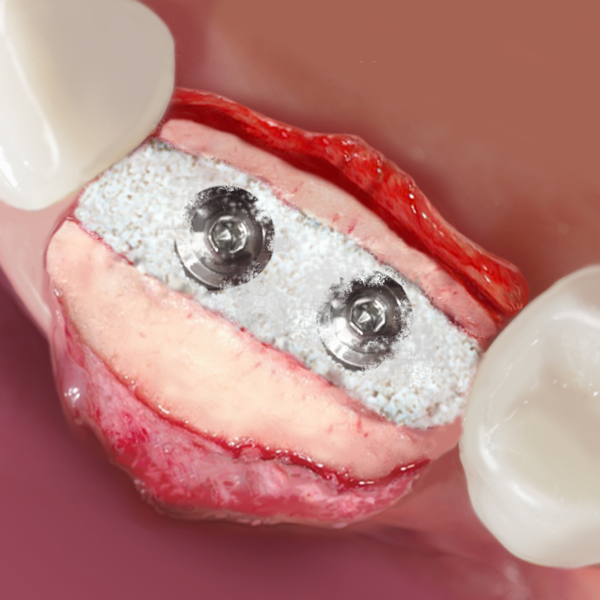
- Lamina barriers are made of cortical bone of heterologous origin produced with an exclusive Tecnoss® process that avoids the ceramization of hydroxyapatite crystals, thus allowing gradual resorption. After a process of superficial decalcification, Lamina soft acquires an elastic consistency, nevertheless maintaining the typical compactness of the bone tissue from which it originates; the margins are soft in order not to cause micro-traumas to the surrounding tissues. Curved soft Lamina has a semi-rigid consistency and should be grafted without hydration, provided that it is previously shaped to fit the defect morphology. Rigid Lamina undergoes a process of superficial semi-decalcification (50% vs Lamina soft) therefore increasing its consistency, typical of the cortical bone tissue(1-2).
- Handling
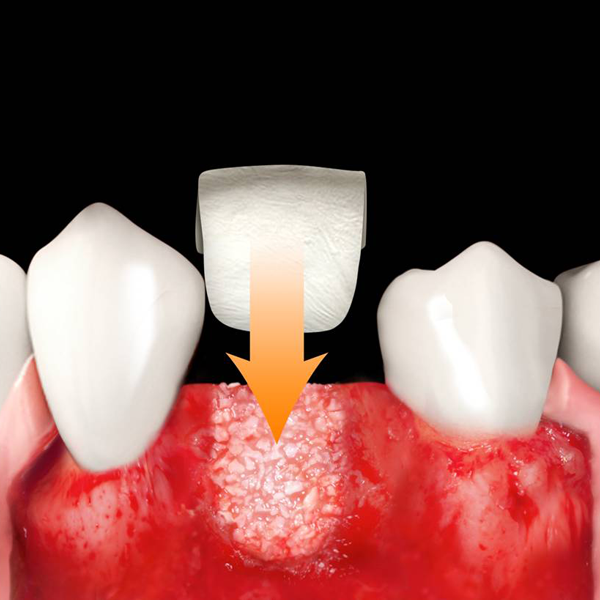
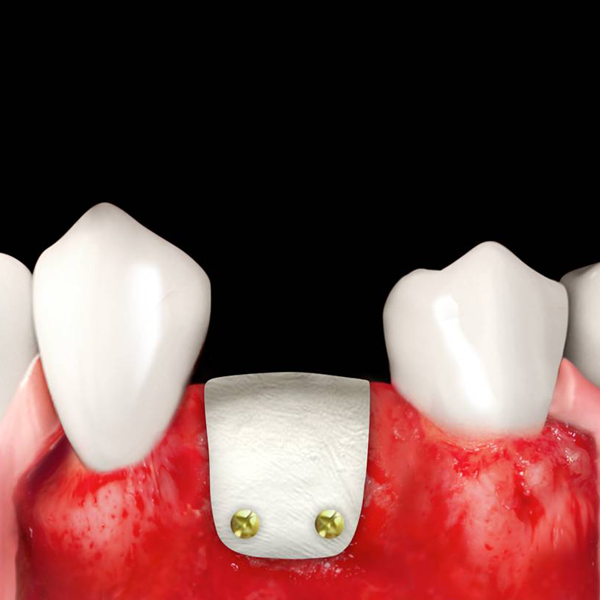
- Lamina soft soft can be shaped with sterile scissors until the desired size is reached, then it must be hydrated for 5/10 minutes in sterile physiological solution. Once it acquires the desired plasticity, it must be adapted to the grafting site; it should always be immobilized either with titanium microscrews or sutured (fine model) directly to the surrounding tissues with a triangular section non-traumatic needle. Curved soft Lamina should not be hydrated in order to maintain its tenting effect but can also be shaped with sterile scissors, and must be fixated with osteosynthesis screws. In case of exposure, Lamina soft should only be removed if there is a clear suprainfection, because its consistency allows to achieve a complete second intention healing of the wound.
- Clinical information
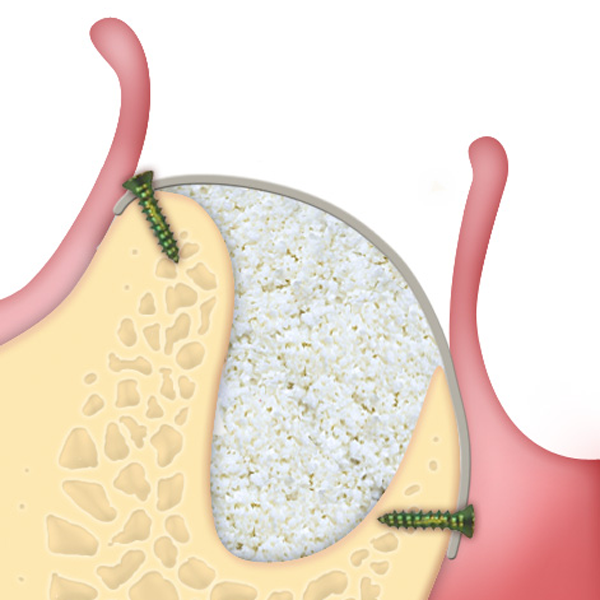
- Lamina soft becomes flexible after hydration and can be shaped(3) and adapted to the defect morphology creating, once fixated with osteosynthesis screws, a semi-rigid covering to the underlying graft(4-6). This property is particularly useful when it is necessary to maintain the graft volume in aesthetic areas, as well as in horizontal augmentation(6-8) of two wall defects and in lateral access sinus lift procedures(5,9,10). Lamina can also be used in regenerations with risks of exposure. Curved soft Lamina has a ~1.0 mm thickness and must be directly grafted without hydration(11): it can be particularly effective in association with GTO® for regeneration of ridges with compromised buccal plate. Rigid Lamina (ref. LS35LS) has been documented for orbital floor and wall reconstruction(12).
The new 0.7 mm thickness Rigid Lamina (ref. LS15LS) represents a viable alternative to autogenous cortical bone plates in the reconstruction of three-dimensional crestal defects with the shell technique.
The above clinical information is based on the experience of expert surgeons - Bibliography
- 1 DI CARLO R, ZARA S, VENTRELLA A, SIANI G, DA ROS T, IEZZI G, CATALDI A, FONTANA A
COVALENT DECORATION OF CORTICAL MEMBRANES WITH GRAPHENE OXIDE AS A SUBSTRATE FOR DENTAL PULP STEM CELLS
NANOMATERIALS, 2019;9:604
2 CABALLÉ-SERRANO J, MUNAR-FRAUA A, DELGADO L, PÉREZ R, HERNÁNDEZ-ALFARO F
PHYSICOCHEMICAL CHARACTERIZATION OF BARRIER MEMBRANES FOR BONE REGENERATION
J MECH BEHAV BIOMED, 2019;97:13-20
3 ROSSI R, RANCITELLI D, POLI PP, RASIA DAL POLO M, NANNMARK U, MAIORANA C
THE USE OF A COLLAGENATED PORCINE CORTICAL LAMINA IN THE RECONSTRUCTION OF ALVEOLAR RIDGE DEFECTS. A CLINICAL AND HISTOLOGICAL STUDY
MINERVA STOMATOL, 2016 OCT;65(5):257-68
4 PAGLIANI L, ANDERSSON P, LANZA M, NAPPO A, VERROCCHI D, VOLPE S, SENNERBY L
A COLLAGENATED PORCINE BONE SUBSTITUTE FOR AUGMENTATION AT NEOSS IMPLANT SITES: A PROSPECTIVE 1-YEAR MULTICENTER CASE SERIES STUDY WITH HISTOLOGY
CLIN IMPLANT DENT RELAT RES, 2012 OCT;14(5):746-58
5 FESTA VM, ADDABBO F, LAINO L, FEMIANO F, RULLO R
PORCINE-DERIVED XENOGRAFT COMBINED WITH A SOFT CORTICAL MEMBRANE VERSUS EXTRACTION ALONE FOR IMPLANT SITE DEVELOPMENT: A CLINICAL STUDY IN HUMANS
CLIN IMPLANT DENT AND RELAT RES, 2013 OCT;15(5):707-13
6 WACHTEL H, FICKL S, HINZE M, BOLZ W, THALMAIR T
THE BONE LAMINA TECHNIQUE: A NOVEL APPROACH FOR LATERAL RIDGE AUGMENTATION - A CASE SERIES
INT J PERIODONTICS RESTORATIVE DENT, 2013 JUL-AUG;33(4):491-7
7 LOPEZ MA, ANDREASI BASSI M, CONFALONE L, CARINCI F, ORMIANER Z, LAURITANO D
THE USE OF RESORBABLE CORTICAL LAMINA AND MICRONIZED COLLAGENATED BONE IN THE REGENERATION OF ATROPHIC CRESTAL RIDGES: A SURGICAL TECHNIQUE. CASE SERIES
J BIOL REGUL HOMEOST AGENTS, 2016 APR-JUN;30(2 SUPPL 1):81-85
8 POLIS-YANES C, CADENAS-SEBASTIÁN C, GUAL-VAQUÉS P, AYUSO-MONTERO R, MARÍ-ROIG A, LÓPEZ-LÓPEZ J
GUIDED BONE REGENERATION OF AN ATROPHIC MAXILLA USING HETEROLOGOUS CORTICAL LAMINA
CASE REP DENT., 2019; 5216362
9 HINZE M, VRIELINCK L, THALMAIR T, WACHTEL H, BOLZ W
ZYGOMATIC IMPLANT PLACEMENT IN CONJUCTION WITH SINUS BONE GRAFTING: THE “EXTENDED SINUS ELEVATION TECHNIQUE”. A CASE-COHORT STUDY
ORAL CRANIOFAC TISSUE ENG, 2011;1:188–197
10 SCARANO A, MURMURA G, MASTRANGELO F, LORUSSO F, GRECO LUCCHINA A, CARINCI F
A NOVEL TECHNIQUE TO PREVENT SINUS MEMBRANE COLLAPSE DURING MAXILLARY SINUS FLOOR AUGMENTATION WITHOUT BONE GRAFT: TECHNICAL NOTE
J BIOL REGUL HOMEOST AGENTS, 2018 NOV-DEC;32(6):1589-1592
11 ROSSI R, FOCE E, SCOLAVINO S
THE CORTICAL LAMINA TECHNIQUE: A NEW OPTION FOR ALVEOLAR RIDGE AUGMENTATION. PROCEDURE, PROTOCOL, AND CASE REPORT
J LEBANESE DENTAL ASS, 2017 JAN-JUN; 52(1):35-41
12 RINNA C, REALE G, FORESTA E, MUSTAZZA MC
MEDIAL ORBITAL WALL RECONSTRUCTION WITH SWINE BONE CORTEX
J CRANIOFAC SURG, 2009 MAY; 20(3):881-4
© 2025 Copyright by Tecnoss Dental Srl. P.I. 08917490016 REA: TO – 1010057. All rights reserved. Web Site developed by DSI Design
Disclosure on personal data processing - Information for customers - Information for suppliers
Disclosure on personal data processing - Information for customers - Information for suppliers
 English
English
 Español
Español
 Italiano
Italiano