Putty



Case Reports
Case Reports
Videos
- Tissue origin :
Cortico-cancellous heterologous bone mix - Tissue collagen :
Preserved plus an additional 20% collagen gel - Composition :
80% granulated mix, 20% collagen gel - Granulometry :
Up to 300 µm - Re-entry time :
About 4 months - Packaging :
Syringe: 0.25 cc, 0.5 cc, 3x0.25 cc, 3x0.5 cc
Wide tip syringe: 1.0 cc - GMDN code : 46425
- Characteristics
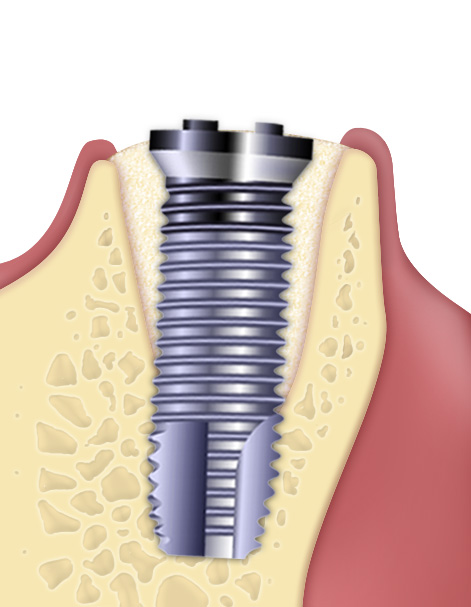
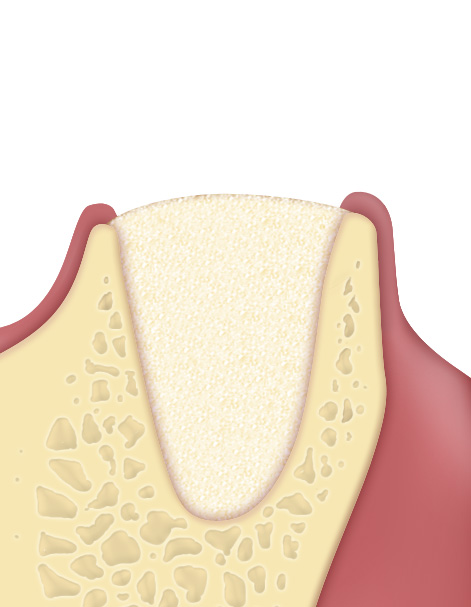
- Putty is a bone paste with at least 80% micronized heterologous bone (granulometry up to 300 µm) and collagen gel. It is made with an exclusive process that provides the product with exceptional malleability and plasticity, making it easy to apply into peri-implant defects with walls. Thanks to its collagen component, the product facilitates blood clotting and the subsequent invasion of repairing and regenerative cells, showing an osteoconductive behaviour(1).
Successful grafting needs complete stability of the biomaterial: for this reason Putty must be used only in cavities able to firmly contain it. Therefore, Putty must not be grafted in two wall defects or in lateral access sinus lift procedures. - Clinical indications
- The extraordinary handling properties of Putty syringe make this product the ideal choice for self-contained peri-implant defects(2) and all small defects that present a self-contained cavity. Furthermore, the Tecnoss® manufacturing process avoids granules ceramization, allowing a progressive resorption of the biomaterial and, at the same time, an adequate new-bone formation rate(3). Putty's “soft” consistency also guarantees an easy and healthy soft-tissues healing.
Thanks to these unique characteristics, Putty has been effectively used for peri-implant defects regeneration: following immediate post-extractive implants placement, Putty can be injected between the defect walls and the implant, guaranteeing a perfect filling of the entire defect volume(4). The product versatility also makes Putty the ideal solution when bone tissue has been lost due to peri-implantitis as long as the containing walls are present. In fact, the primary condition for gaining a successful regeneration is to achieve the biomaterial initial stability. Therefore, Putty must be used only in defects where the surrounding walls guarantee such condition: for example inside the bone crest when ridge-split technique is adopted(5), or with horizontally resorbed crests, in association with OsteoBiol® Lamina (Bone Layer technique)(6). - Bibliography
- (1) ARCURI C, CECCHETTI F, GERMANO F, MOTTA A, SANTACROCE C
CLINICAL AND HISTOLOGICAL STUDY OF A XENOGENIC BONE SUBSTITUTE USED AS A FILLER IN POSTEXTRACTIVE ALVEOLUS
MINERVA STOMATOL, 2005 JUN;54(6):351-62
(2) BARONE A, AMERI S, COVANI U
IMMEDIATE POSTEXTRACTION IMPLANTS: TREATMENT OF RESIDUAL PERI IMPLANT DEFECTS. A RETROSPECTIVE ANALYSIS
EUR J IMPLANT PROSTHODONTICS, 2006,2: 99-106
(3) NANNMARK U, AZARMEHR I
SHORT COMMUNICATION: COLLAGENATED CORTICOCANCELLOUS PORCINE BONE GRAFTS. A STUDY IN RABBIT MAXILLARY DEFECTS
CLIN IMPLANT DENT RELAT RES, 2010 JUN 1; 12(2):161-3
(4) CASSETTA M, RICCI L, IEZZI G, DELL'AQUILA D, PIATTELLI A, PERROTTI V
RESONANCE FREQUENCY ANALYSIS OF IMPLANTS INSERTED WITH A SIMULTANEOUS GRAFTING PROCEDURE: A 5-YEAR FOLLOW-UP STUDY IN MAN
INT J PERIODONTICS RESTORATIVE DENT, 2012 OCT;32(5):581-9
(5) SANTAGATA M, GUARINIELLO L, TARTARO G
A MODIFIED EDENTULOUS EXPANSION (MERE) TECHNIQUE FOR IMMEDIATE PLACEMENT OF IMPLANTS. A CASE REPORT
J ORAL IMPLANTOL, 2011 MAR;37 SPEC N.:114-9
(6) LOPEZ MA, ANDREASI BASSI M, CONFALONE L, CARINCI F, ORMIANER Z, LAURITANO D
THE USE OF RESORBABLE CORTICAL LAMINA AND MICRONIZED COLLAGENATED BONE IN THE REGENERATION OF ATROPHIC CRESTAL RIDGES: A SURGICAL TECHNIQUE. CASE SERIES
J BIOL REGUL HOMEOST AGENTS, 2016 APR-JUN;30(2 SUPPL 1):81-85
© 2025 Copyright by Tecnoss Dental Srl. P.I. 08917490016 REA: TO – 1010057. All rights reserved. Web Site developed by DSI Design
Disclosure on personal data processing - Information for customers - Information for suppliers
Disclosure on personal data processing - Information for customers - Information for suppliers
 English
English
 Español
Español
 Italiano
Italiano